Early-stage multiple myeloma and high-risk smoldering myeloma raise an important clinical question: when should active treatment begin, and which option offers safer long-term control? Patients and families often search online for clear answers while consulting a cancer doctor in Vadodara who can guide them with data, not opinions. Two major clinical trials—AQUILA and QuiReDEX—have shaped current discussion. Each trial answers a different need, and the choice depends on disease risk, patient profile, and future treatment goals.
This page explains both studies in simple terms and clarifies why modern practice now leans toward AQUILA in many cases.
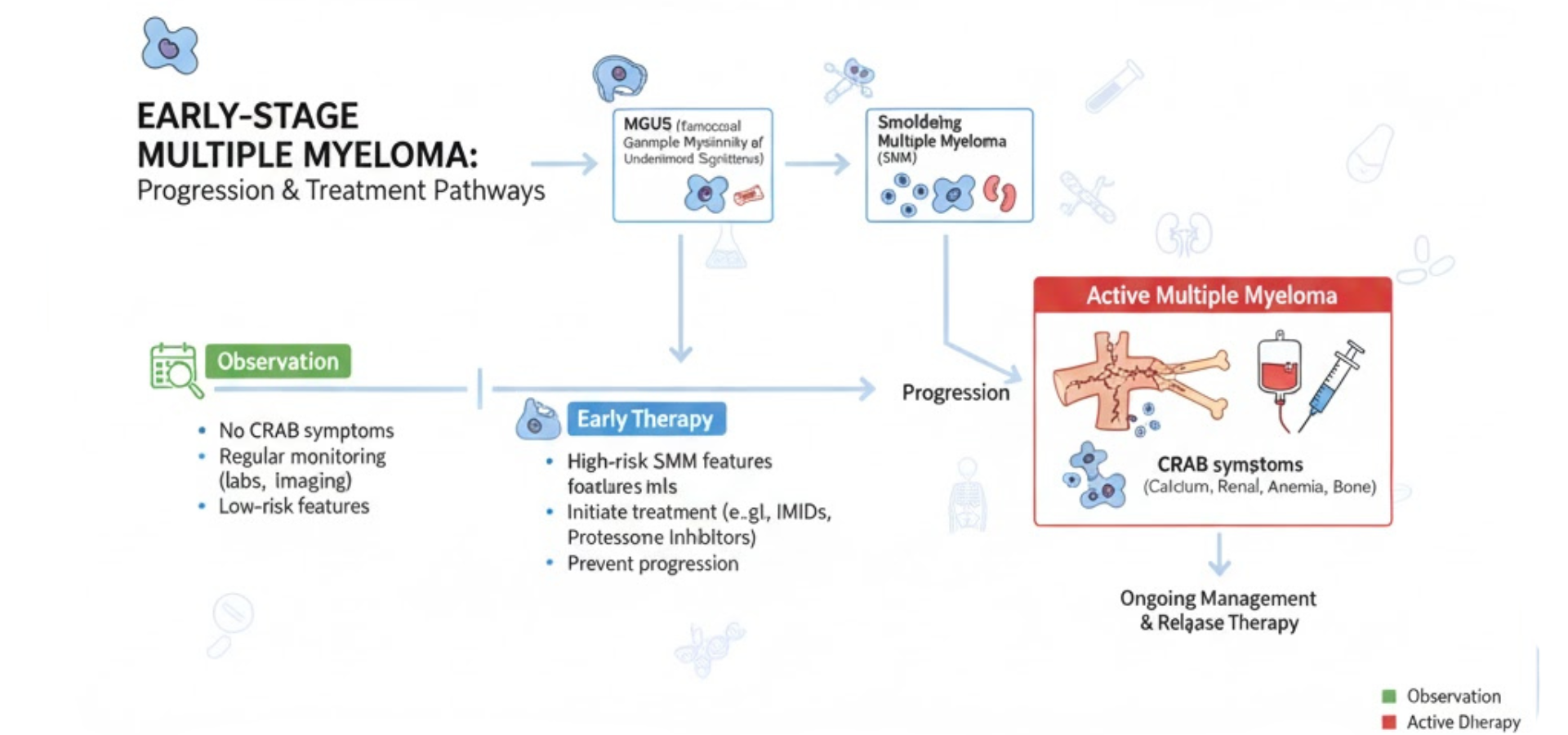
Why Early Treatment Matters in High-Risk Smoldering Myeloma
Smoldering myeloma sits between MGUS and active multiple myeloma. Some patients remain stable for years, while others progress fast and face bone damage, kidney issues, anemia, or high calcium levels. Delayed care may result in emergency treatment once CRAB or SLiM criteria appear.
Many people search for a cancer doctor near me when symptoms start late. Current oncology care focuses on delaying progression before organ damage begins, while also avoiding unnecessary toxicity.
The AQUILA Trial: Daratumumab as Early Intervention
AQUILA studied daratumumab monotherapy in high-risk smoldering myeloma compared with active observation.
Key Outcomes from AQUILA
- Higher response rate: 63.4% with daratumumab vs 2.0% with monitoring
- Delayed need for full treatment: Only 33% of patients on daratumumab required active myeloma therapy, compared with 52% under observation
- Lower CRAB-based progression: 12 patients vs 34 patients
- Lower SLiM-based progression: 50 patients vs 65 patients
- Second progression-free survival: 42% risk reduction
- Overall survival benefit: 60-month survival of 93.0% vs 86.9%
These results show clear disease control without immediate exposure to multi-drug chemotherapy.
Why AQUILA Changed Practice
Daratumumab targets CD38 on plasma cells and works as a single agent. This approach reduces early toxicity, preserves future treatment options, and lowers organ damage risk. For many oncologists, this balance fits patients who want control without aggressive regimens.
Why Len-Dex Was Not the Comparator in AQUILA
A common question remains: Why was lenalidomide plus dexamethasone not used as the comparison arm?
The answer lies in trial design ethics and clinical reality.
Len-Dex already showed benefit in earlier trials. Using it as a comparator would expose patients to known side effects such as infections, blood clots, fatigue, and steroid-related complications. AQUILA aimed to test whether a gentler option could delay disease while keeping quality of life stable. Observation was the ethical control arm for that purpose.
The QuiReDEX Trial: Lenalidomide plus Dexamethasone
QuiReDEX evaluated lenalidomide with dexamethasone versus observation in high-risk smoldering myeloma.
Key Outcomes from QuiReDEX
- Time to progression improved: Median time not reached with treatment vs 23 months with observation
- Lower conversion to active myeloma: 39% with treatment vs 86% under observation
- Overall survival: Median not reached in either group
- Post-progression survival: Similar in both groups after later treatments
This trial confirmed that early therapy can delay progression. However, it also highlighted long-term exposure to steroids and immunomodulatory drugs.
AQUILA vs QuiReDEX: Practical Comparison
Aspect | AQUILA | QuiReDEX |
Treatment type | Single-agent antibody | Dual drug + steroid |
Toxicity burden | Lower | Moderate |
Progression delay | Strong | Strong |
Survival trend | Positive | Neutral |
Quality of life focus | High | Moderate |
Future therapy preservation | Better | Limited |
Which Option Fits Today’s Patients?
Modern oncology values disease control, safety, and long-term planning. AQUILA suits patients who need early control with fewer side effects. QuiReDEX remains relevant where aggressive risk demands faster cytoreduction and where patients tolerate combination therapy well.
A cancer doctor in Vadodara evaluates age, kidney status, bone health, infection risk, and patient preference before advising either path.
Role of Specialist Care in Decision Making
Online data alone cannot guide therapy. A trained oncologist reviews imaging, marrow reports, cytogenetics, and blood markers. At Dr. Mrugal Doshi’s oncology practice in Vadodara, treatment plans follow evidence-based protocols with transparent discussion.
Patients often search for an oncologist in Vadodara who explains choices clearly. Trust, follow-up, and ethical guidance remain central to care.
Current Oncology Direction
Recent global guidelines lean toward early but measured intervention. AQUILA supports this shift by showing that targeted therapy can delay disease and extend survival without early chemotherapy exposure.
Final Takeaway
- QuiReDEX proved early treatment works
- AQUILA proved early treatment can be safer and longer lasting
- Len-Dex was not a comparator due to toxicity concerns and trial ethics
- Daratumumab now stands as a preferred early option for many high-risk patients
For patients facing smoldering myeloma decisions, consultation with an experienced cancer doctor in Vadodara ensures the right balance between control and quality of life.
Frequently Asked Questions (FAQs)
What is smoldering multiple myeloma?
Smoldering multiple myeloma is an early stage of myeloma where cancer cells are present but symptoms like bone damage or kidney problems are not yet seen.
When should treatment start for smoldering myeloma?
Treatment is advised in high-risk cases where there is a strong chance of progression to active multiple myeloma within a short time.
What is the AQUILA trial in multiple myeloma?
The AQUILA trial studied daratumumab as early treatment in high-risk smoldering myeloma and showed better disease control and survival outcomes.
What is daratumumab used for?
Daratumumab is a targeted cancer medicine used in myeloma to control abnormal plasma cells and delay disease progression.
How is AQUILA different from QuiReDEX?
AQUILA used single-drug therapy with daratumumab, while QuiReDEX used lenalidomide and dexamethasone combination treatment.
Why was lenalidomide-dexamethasone not compared in AQUILA?
Len-dex has known side effects. AQUILA aimed to test a safer early option, so observation was used as the comparison group.
What are CRAB and SLiM criteria in myeloma?
These are medical signs used to decide when smoldering myeloma becomes active and needs full treatment.
Which treatment has fewer side effects, AQUILA or QuiReDEX?
AQUILA showed fewer side effects because it uses single-agent targeted therapy instead of combination drugs with steroids.
Can early treatment improve survival in myeloma?
Yes, studies like AQUILA show that early treatment can delay progression and improve long-term survival.
Which doctor should I consult for multiple myeloma in Vadodara?
You should consult an experienced oncologist. Dr. Mrugal Doshi is a cancer doctor in Vadodara who provides evidence-based myeloma care.